PROVIDENCE, R.I. [Brown University] — Over the last few years, a class of compounds called ADEPs (cyclic acyldepsipeptides) has emerged as a promising new weapon in the fight against drug-resistant bacteria. The compounds work by attaching themselves to a cellular enzyme called ClpP, which bacterial cells use to rid themselves of harmful proteins. With an ADEP attached, ClpP can’t function properly, and the bacterial cell dies.
Now scientists from Brown University and Massachusetts Institute of Technology have shown new details of how ADEPs bind to the ClpP complex in the bacterium that causes tuberculosis. The researchers are hopeful that the work, published in Proceedings of the National Academy of Sciences, could serve as a blueprint for optimizing ADEPs for treatment of tuberculosis.
Jason Sello, associate professor of chemistry at Brown and a co-author of the new study, refers to ClpP as a “cellular garbage disposal.” The ClpP complex is a barrel-shaped molecule that devours and degrades any harmful proteins that might be floating around within the cell. ClpP works with a set of partner proteins, including one called ClpX. ClpX gathers up unwanted proteins, opens up a hole in the ClpP barrel, and feeds the offending proteins through to be degraded.
ADEPs are thought disrupt this process by posing as a ClpX mimic. As imposters, the ADEPs bind to ClpP in the same spot that ClpX normally would. With an ADEP attached, ClpP runs amok, devouring proteins indiscriminately and eventually destroying the cell. It’s through this process that ADEPs have been shown in lab experiments to kill several types of harmful bacterial, including S. aureus and S. pneumoniae.
The compounds aren’t nearly as effective, however, against Mycobacterium tuberculosis (Mtb) and Sello and his colleagues wanted find out why. One potential explanation was that the ClpP complex in Mtb is much more complicated than it is in other bacteria.
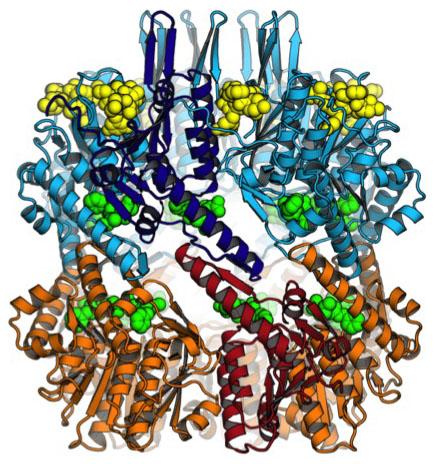
In most bacteria, the ClpP barrel is an assembly of 14 copies of a single protein. But in Mtb, it’s made up of seven copies of two different proteins, called ClpP1 and ClpP2. What’s more, previous work had suggested that ClpP1 and ClpP2 could only form an active complex in the presence of a partner like ClpX, as well as a protein destined for destruction by ClpP.
“The resistance of the Mycobacterium tuberculosis to the ADEPs and the peculiarity of its ClpP piqued our curiosity,” said Sello, who worked on the research with a group of MIT biochemists led by Robert Sauer. “We wanted to determine if the ADEPs could bind and perturb the structure and function as they do in other bacteria.”
To do that, the researchers mixed the two ClpP proteins from Mtb in a test tube. They then added ADEPs as a mimic for ClpX, and a compound called a peptide agonist that mimics a protein to be degraded. As they hoped, the researchers showed that the concoction did indeed yield an active ClpP complex capable of degrading both peptides and proteins.
That in itself was an interesting finding, Sello says. It confirms the idea that the ADEPs are able to mimic the ClpX and induce ClpP1, ClpP2, and the agonist to come together to form a hyperactive ClpP complex. They also discovered that novel ADEP analogs, synthesized in Sello’s lab and used in the experiments, were better at binding and activating ClpP than those that were previously reported.
“From the perspective of drug discovery, these results were important because they showed that the ADEPs could affect the function of ClpP in Mtb as they do in other bacteria and that the potency of the ADEPs could be improved,” Sello said.
From there, the researchers went a step further. They used X-ray crystallography to analyze the binding of optimized ADEPs to ClpP from Mtb at atomic resolution. The team was surprised to find that the ADEPs bind only to the ClpP2 proteins in the complex, not to ClpP1. That could be a reason ADEPs aren’t as effective against TB, Sello says.
“Fortunately, the structure provides strategies for designing ADEPs that bind better to ClpP,” Sello said. “We have already generated many new ideas about how the ADEP structure can be rationally modified to improve ClpP binding and thus killing of M. tuberculosis. We are very optimistic that some of our designs could ultimately be a new class of drugs for the treatment of tuberculosis.”
Sello and his team have already begun experiments to see if their new ideas are viable. The group is hopeful that this detailed picture of ADEPs bound to ClpP will continue to reveal new approaches to fighting TB.
The research paper was co-authored by Karl Schmitz, a post-doctoral researcher at MIT, and Daniel Carney, who earned his Ph.D. in Sello’s lab at Brown. The work was supported by the National Institutes of Heath (GM-101988) and Brown University. Jason Sello is the recipient of a CAREER award from the National Science Foundation.