PROVIDENCE, R.I. [Brown University] — Our cells are more like us than we may think. They’re sensitive to their environment, poking and prodding deliberately at their surroundings with hand-like feelers and chemical signals as they decide whether and where to move. Such caution serves us well but has vexed engineers who seek to create synthetic tissue, heart valves, implants and other devices that the human body will accept.
To overcome that obstacle, scientists have sought to learn more about how cells explore what’s around them. While numerous studies have looked at cellular movement in two dimensions and a few recent experiments involved cellular motion in three dimensions, scientists remained unsure just how much cells interacted with their surroundings. Now, a study involving Brown University and the California Institute of Technology has recorded for the first time how cells move in three dimensions by measuring the force exerted by cells on their environs. The research gives scientists their most complete assessment to date about how cells move.
“We’ve learned that cells move in much more complex ways than previously believed,” said Christian Franck, assistant professor in engineering at Brown and the co-lead author of the study published online in the Proceedings of the National Academy of Sciences. “Now, we can start to really put numbers on how much cells push and pull on their environment and how much cells stick to tissues as they move around and interact.”
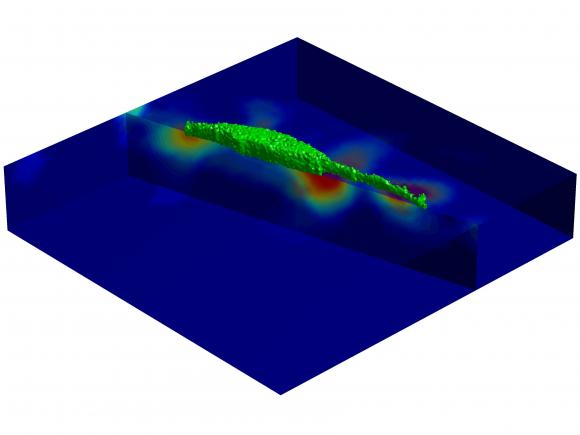
In the study, Franck and co-lead author Stacey Maskarinec, who both conducted the experiments while graduate students at the California Institute of Technology, placed cells on top of a 50-micron-thick water-based gel designed to mimic human tissue. They added into the gel spheres about a half-micron in diameter that lit up when jostled by the cells’ actions. By combining two techniques — laser scanning confocal microscopy and digital volume correlation — the scientists tracked the cells’ movement by quantifying exactly how the environment changed each time the cell moved. The team recorded results every 35 minutes over a 24-hour period.
What they found was cells move in intriguing ways. In one experiment, a cell is clearly shown operating in three dimensions by extending feelers into the gel, probing at depth, as if thrusting a leg downward in a pool. The Brown and Caltech scientists also found that as a cell moves, it engages in a host of push-pull actions: It redistributes its weight, it coils and elongates its body, and it varies the force with which it “grips,” or adheres, to a surface. Combined, the actions help the cell generate momentum and create a “rolling motion,” as Franck described it, that is more like walking than shuffling, as many scientists had previously characterized the movement.
“The motion itself is in three dimensions,” Franck said.
“The method that Christian and Stacey developed is unique in that it allows them to look directly at changes in force over time,” said David Tirrell, the Ross McCollum-William H. Corcoran Professor and professor of chemistry and chemical engineering at Caltech. “If they want to look at the overall forces, they can do that, too, by allowing the cells to relax and then comparing images before and after relaxation of the stress.”
Franck’s lab plans to use the new force-measurement technique to examine how the movement of normal healthy cells differs from mutant or malignant ones. “That promises to give us greater insight in how cells’ behavior changes when they become diseased,” Franck said.
Tirrell and Guruswami Ravichandran, scientists at Caltech, contributed to the research. The National Science Foundation funded the work.